30/04: EU Monitor – Participate in the EAHP-EPSA Student Science Award!
The EAHP EU Monitor is a regular round up of news relevant to hospital pharmacy in Europe.
You can subscribe to receive the EAHP EU Monitor by email HERE.
Students sign up for the EAHP-EPSA Student Science Award!

The European Association of Hospital Pharmacists (EAHP) is proud to announce the opening of the next round of the EAHP-EPSA Student Science Award. This prestigious prize recognises and honours the best scientific research authored by a pharmacy student or recent graduate. Since 2011 the EAHP-EPSA Student Science Award is offered to one member of the European Pharmaceutical Students' Association (EPSA) who has conducted research in the field of hospital and/or clinical pharmacy.
To enter into the competition, students and recent graduates are required to attend the webinar “How to write an abstract”, hosted by Nenad Miljković, EAHP Director of Professional Development, on 4th June 2019. Participation in the webinar is obligatory for students wishing to compete for the 2020 EAHP-EPSA Student Science Award. Registration closes on 31st May 2019.
After the webinar, participating students and recent graduates can obtain detailed feedback on their work prior to submitting their final abstract by 15th November. The abstracts are reviewed in accordance with the criteria of innovation, originality, and contribution to the development of hospital pharmacy. The winner will be announced during EAHP’s 25th Congress.
Register HERE for the webinar “How to write an abstract”
More information about the EAHP-EPSA Student Science Award HERE
1st WHO guideline on digital health interventions

In mid-April, the World Health Organisation (WHO) released a guideline containing recommendations on digital interventions for health system strengthening. The document includes information on 10 ways that countries can use digital health technology, accessible via mobile phones, tablets and computers, to improve people’s health and essential services. It also provides a better understanding of which digital health interventions have an evidence base to address health system needs and targets decision-makers in health ministries, public health practitioners and other stakeholders.
In relation to health workers, the guideline stresses the need for adequate training to boost their motivation to transition towards working more easily with emerging digital technologies. In particular supportive environments for training, dealing with unstable infrastructure, as well as policies to protect the privacy of individuals need to be provided.
Read the guidelines HERE
New AMR report calls for urgent action
The Interagency Coordination Group on antimicrobial resistance (IACG) – which includes members from the United Nations (UN), the World Health Organization (WHO), the Food and Agriculture Organization and the World Organisation for Animal Health – released a report containing critical recommendations to combat drug-resistant infections and to prevent the staggering number of deaths caused by antimicrobial resistance each year.
Antimicrobial resistance (AMR) is increasingly becoming a global problem which calls for immediate, coordinated and ambitious action. In line with the European Commission’s One Health initiative, also the IACG report calls for a multisectoral “One Health” approach in order to jointly avert a potentially disastrous drug-resistance crisis.
Recommendations of the IACG are linked to the acceleration of progress, innovation, collaboration, investment into sustainable responses as well as strengthened accountability and global governance. Countries are encouraged
- to prioritize national action plans to scale-up financing and capacity-building efforts;
- to put in place stronger regulatory systems and support awareness programs for responsible and prudent use of antimicrobials by professionals in human, animal and plant health;
- to invest in ambitious research and development for new technologies to combat antimicrobial resistance; and
- to urgently phase out the use of critically important antimicrobials as growth promoters in agriculture.
The report calls on countries to intensify the efforts and to jointly work together on overcoming AMR which has also been identified as a major barrier to the implementation of the UN’s Sustainable Development Goals, in particular those linked to universal health coverage, secure and safe food, sustainable farming systems and clean water and sanitation.
Access the report HERE
Special Eurobarometer on vaccination
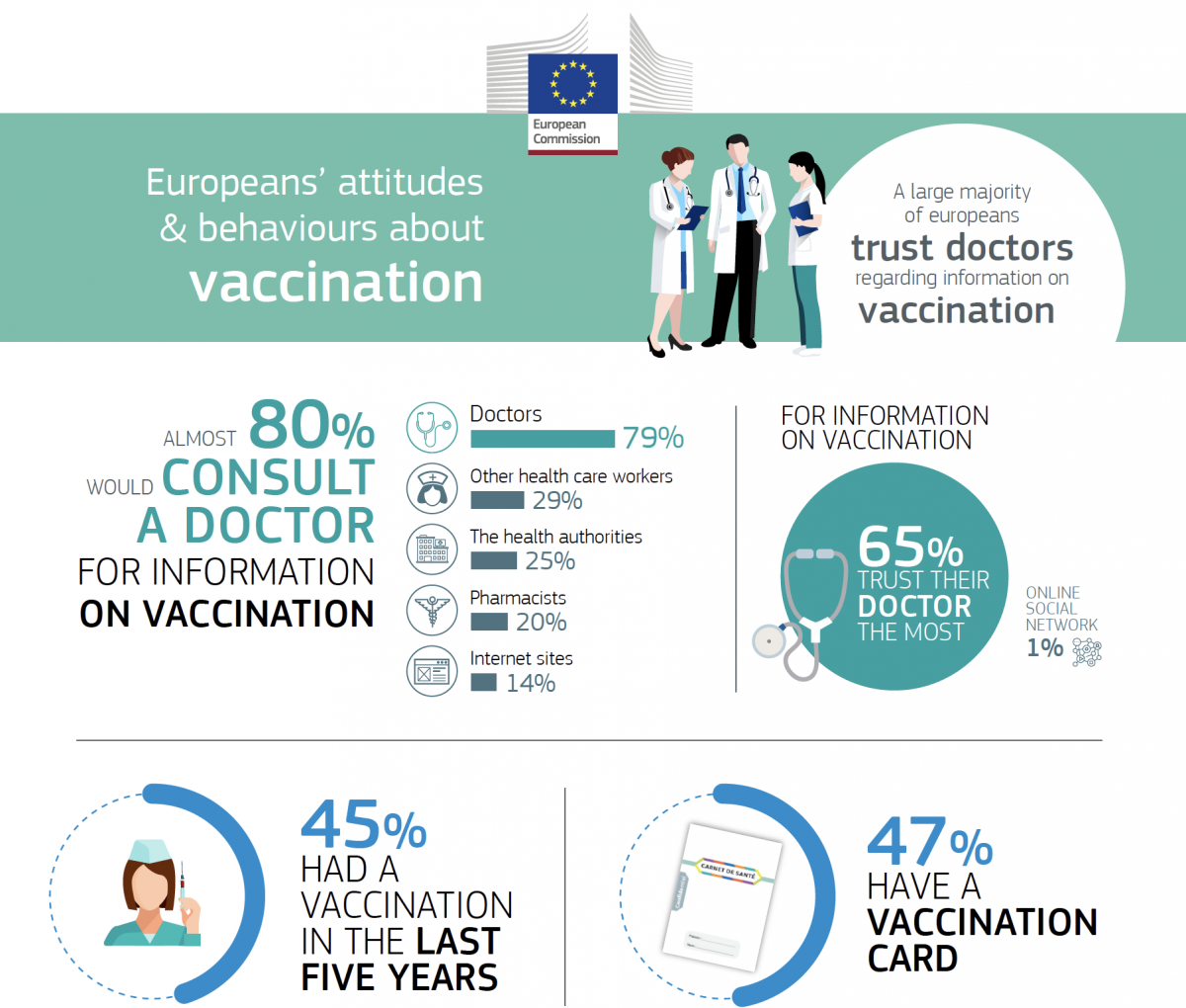
During the 2019 European Immunisation Week a Special Eurobarometer was released detailing information on Europeans’ attitudes towards vaccination. Data gathered shows that 85% of EU citizens believe vaccination is an effective way to prevent infectious diseases and to protect oneself and others. Around half of the EU citizens have been vaccinated in the last five years and a large majority (79%) consult and trust a healthcare professional, such as their pharmacist, to get information about vaccinations.
Information gathered via the Special Eurobarometer forms part of an elaborate European Commission initiative which addresses vaccine disinformation and seeks to increase vaccine coverage across the EU. Together with the WHO, the European Commission will be hosting a Global Vaccination Summit in September 2019 in order to give high level visibility and political endorsement to the topic of vaccination.
Access the Special Eurobarometer HERE
EJHP: Quality and safety of parenteral nutrition for newborn and preterm infants as an on-ward preparation

The online first edition of the European Journal of Hospital Pharmacy (EJHP) recently published an original article addressing the quality and safety of parenteral nutrition for newborn and preterm infants as an on-ward preparation. The study concluded that component concentrations of parenteral nutrition prepared on wards by nurses differed frequently and significantly from their medical prescription, and the deviation can be critical depending on the component and its mode of action.
Read article HERE
Have you already set up your account to read the EJHP online?
Hospital pharmacists affiliated with one of EAHP’s member associations have free access to the EJHP. In order to gain access to the journal online, members of national hospital pharmacy associations that are also members of EAHP have to set up a user account via the following LINK. The registration manual outlines on the one hand how a user account can be set up and on the other hand how the EJHP can be accessed via EAHP’s website.
For more information or in case of questions please contact info[at]eahp[dot]eu.
Are you ready for the Statement Implementation Month?
Watch EAHP’s social media channels in May to follow the activities of the Statement Implementation Month. In the coming weeks, EAHP will be sharing interesting facts and figures about the European Statements of Hospital Pharmacy that express commonly agreed objectives which every European health system should aim for in the delivery of hospital pharmacy services. Visit EAHP’s Facebook, Twitter, LinkedIn and Instagram pages or check out the Statements website to learn more about the project.
Learn more about SAT HERE
------------------------------------------------------------------------------------------------

Consultations
Public consultation on EMA Regulatory Science to 2025
The purpose of this public consultation is to seek views from EMA’s stakeholders, partners and the general public on EMA’s proposed strategy on Regulatory Science to 2025 and whether it meets stakeholders’ needs. By highlighting where stakeholders see the need as greatest, participants have the opportunity to jointly shape a vision for regulatory science that will in turn feed into the wider EU network strategy in the period 2020-25.
Deadline – 30th June 2019
Access consultation HERE
EMA - Guideline on the evaluation of medicinal products indicated for treatment of bacterial infections
The EMA has launched a consultation on the revision of its guideline on the evaluation of human medicines indicated for the treatment of bacterial infections. Antimicrobial resistance is a global public health problem. Regulators in the European Union, the United States and Japan have had extensive discussions over the last few years to explore and agree how to align as much as possible their respective data requirements so that medicine developers can design clinical trials that meet the evidence needs of multiple regulatory agencies. The revised guidance reflects the outcome of these discussions.
Deadline – 31st July 2019
Access consultation HERE
EMA – Public consultation on key principles for the electronic product information of EU medicines
The European Medicines Agency (EMA), together with the European Commission (EC), has launched a public consultation on draft key principles which will form the basis on which the electronic product information (ePI) for human medicines will be developed and used throughout the European Union. The rationale behind the ePI is that digital platforms open additional possibilities to disseminate the PI electronically. This can address some of the current limitations and better meet patients’ and healthcare professionals’ needs for accessible, up-to-date information on medicines. The draft key principles are the result of extensive discussions and consultations carried out by EMA, the Heads of Medicines Agencies (HMA) and the EC throughout 2018, with representatives of all stakeholder groups.
Deadline – 31st July 2019
Access consultation HERE





