29th Congress of the EAHP
Vertical Tabs
Aircraft and airport
Further information via Copenhagen Airport or SAS.
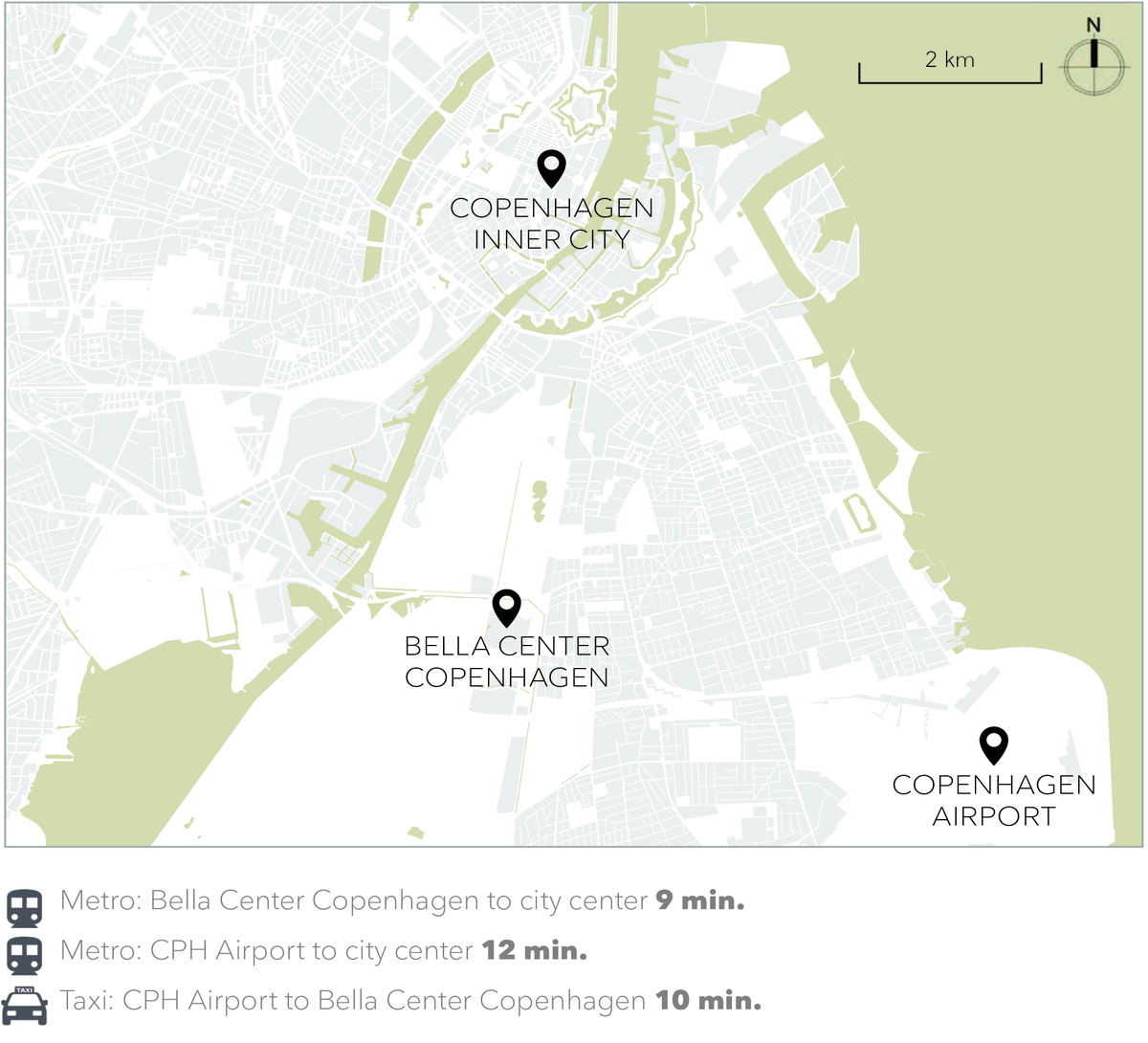
Bella Sky Conference & Event is just a 10 minute taxi drive from Copenhagen Airport. The regional trains run from the airport to Ørestad Station, where you can change to metro M1 (1 stop).
Taxi
A taxi to/from the city center or the airport costs around DKK 200-250 in normal traffic condition
Metro
With the metro you can get to the center of Copenhagen in about 10 minutes. With metro line M1, which runs between Vanløse and Vestamager, you should get off at Bella Center metro station. From the station, walk between AC Hotel Bella Sky Copenhagen's two towers and find entrance 3.
Further information at www.m.dk.
Bus
Several buses stop at the bus stop in front of our door.
Departure times can be found at DOT.
You can also use Rejseplanen to plan your trip by public transport.
Train
Bella Sky Conference & Event is just 10-15 minutes by taxi from Copenhagen Central Station. To get to and from Copenhagen Central Station, you can also take bus 30, which takes 20-25 minutes. All regional trains stop at Ørestad Station, from where you can change to the metro. Please note that S-trains do not stop at Ørestad Station. Find more information and book tickets via DSB.
Car and parking options
There is a motorway almost to the door from both Denmark and Sweden.
Follow "Lufthavnsmotorvejen" with route no. E20, and use exit 19 (Ørestad/Bella Center)
The closest parking is at AC Hotel Bella Sky (P1)
Paid parking is managed by
- APCOA - area code 3994.
- EasyPark – area code 39940
It is also possible to park at the Royal Golf Club on the other side of Center Boulevard.